Processing pipeline details
ASLPrep [1][2] adapts its pipeline depending on what data and metadata are available and are used as inputs. It requires the input data to be BIDS-valid and include necessary ASL parameters.
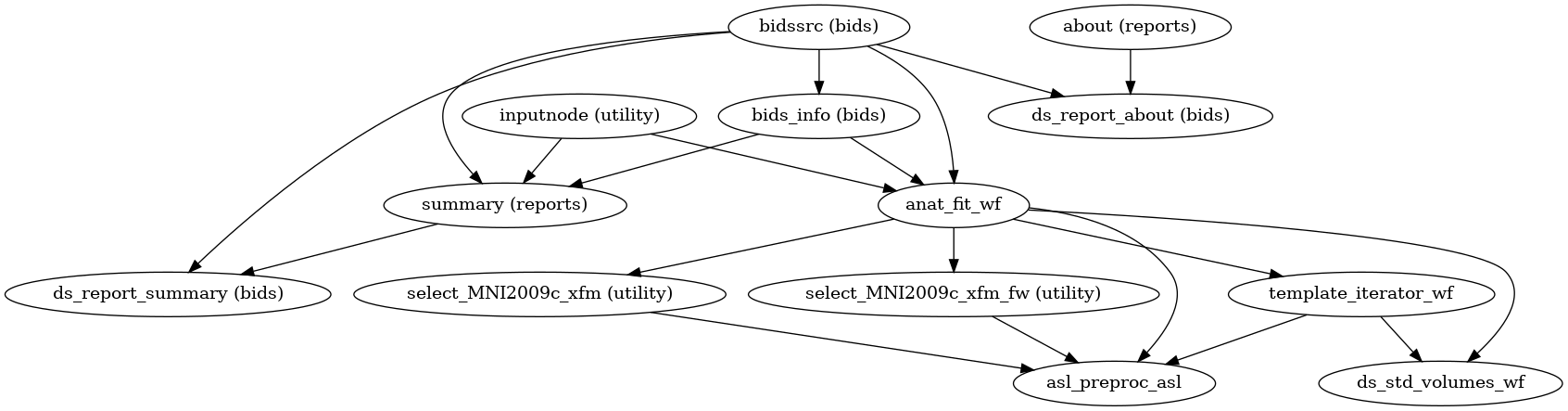
(Source code, png, svg, pdf)
Structural Preprocessing
The anatomical sub-workflow is from sMRIPrep. It first constructs an average image by conforming all found T1w images to a common voxel size, and, in the case of multiple images, averages them into a single reference template.
See also sMRIPrep’s
init_anat_preproc_wf().
Brain extraction, brain tissue segmentation and spatial normalization
Next, the T1w reference is skull-stripped using a Nipype implementation of
the antsBrainExtraction.sh tool (ANTs), which is an atlas-based
brain extraction workflow:
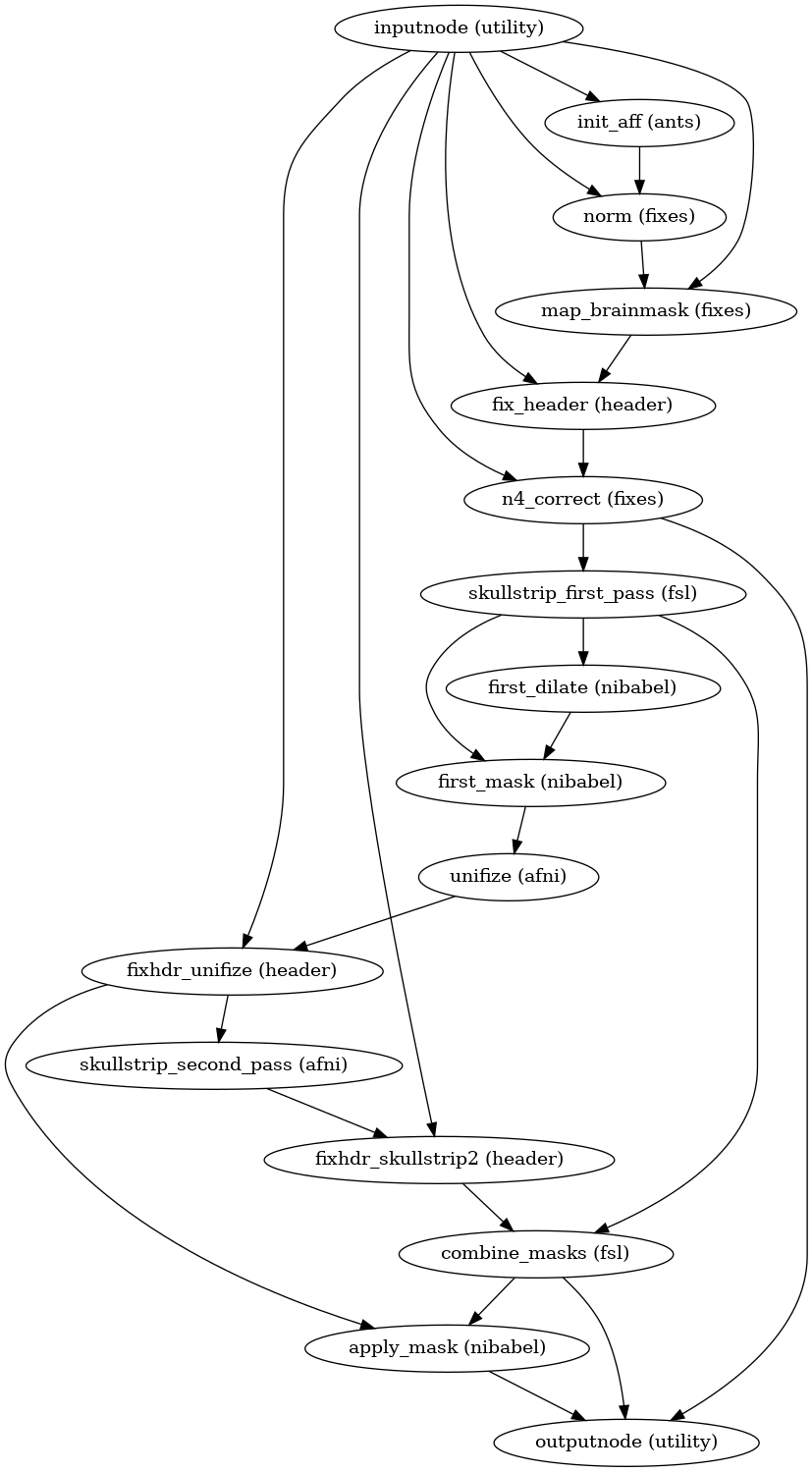
(Source code, png, svg, pdf)
An example of brain extraction is shown below:
Brain extraction
Once the brain mask is computed, FSL fast is used for brain tissue segmentation.
Brain tissue segmentation
Finally, spatial normalization to standard spaces is performed using ANTs’ antsRegistration
in a multiscale, mutual-information based, nonlinear registration scheme.
See Standard and nonstandard spaces for more information on how standard and nonstandard spaces can
be set to resample the preprocessed data onto the final output spaces.
Animation showing spatial normalization of T1w onto the MNI152NLin2009cAsym template.
ASL preprocessing
Preprocessing of ASL files is split into multiple sub-workflows described below.
ASL reference image estimation
init_asl_reference_wf()
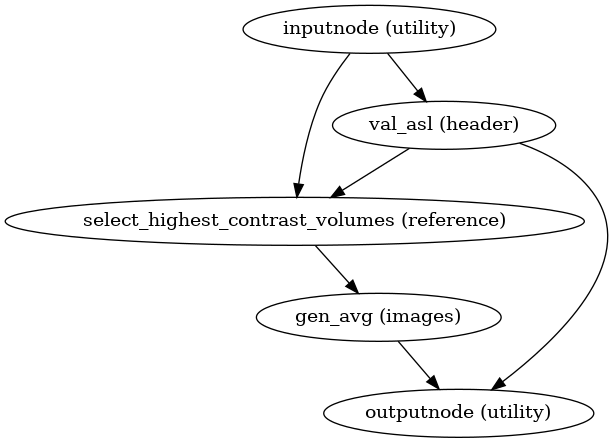
(Source code, png, svg, pdf)
This workflow estimates a reference image for an ASL series.
The reference image is then used to calculate a brain mask for the
ASL signal using NiWorkflow’s
init_enhance_and_skullstrip_bold_wf().
Subsequently, the reference image is fed to the head-motion estimation
workflow and the registration workflow to map the
ASL series onto the T1w image of the same subject.
Calculation of a brain mask from the ASL series.
Head-motion estimation
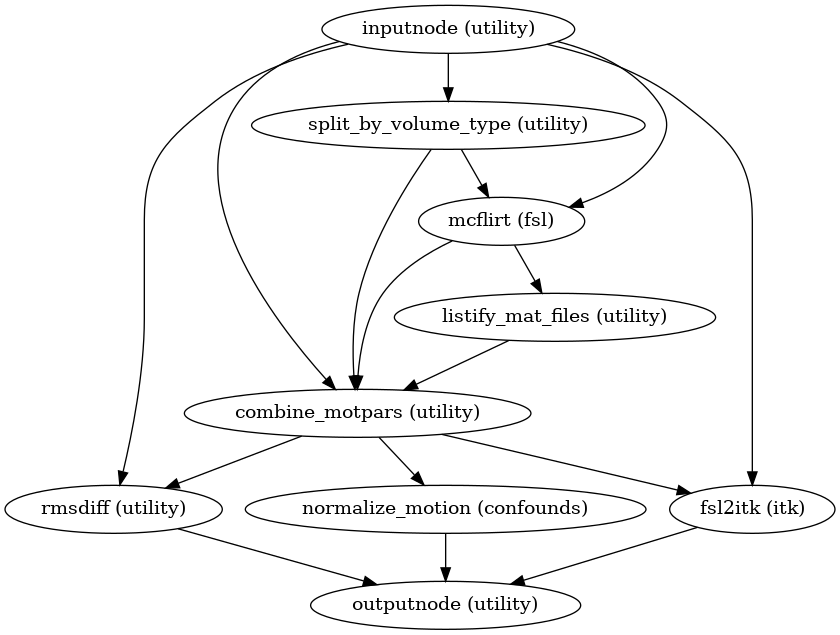
(Source code, png, svg, pdf)
Using the previously estimated reference scan,
FSL mcflirt is used to estimate head-motion.
As a result, one rigid-body transform with respect to
the reference image is written for each ASL
time-step.
Additionally, a list of 6-parameters (three rotations and
three translations) per time-step is written and fed to the
confounds workflow,
for a more accurate estimation of head-motion.
Confounds estimation
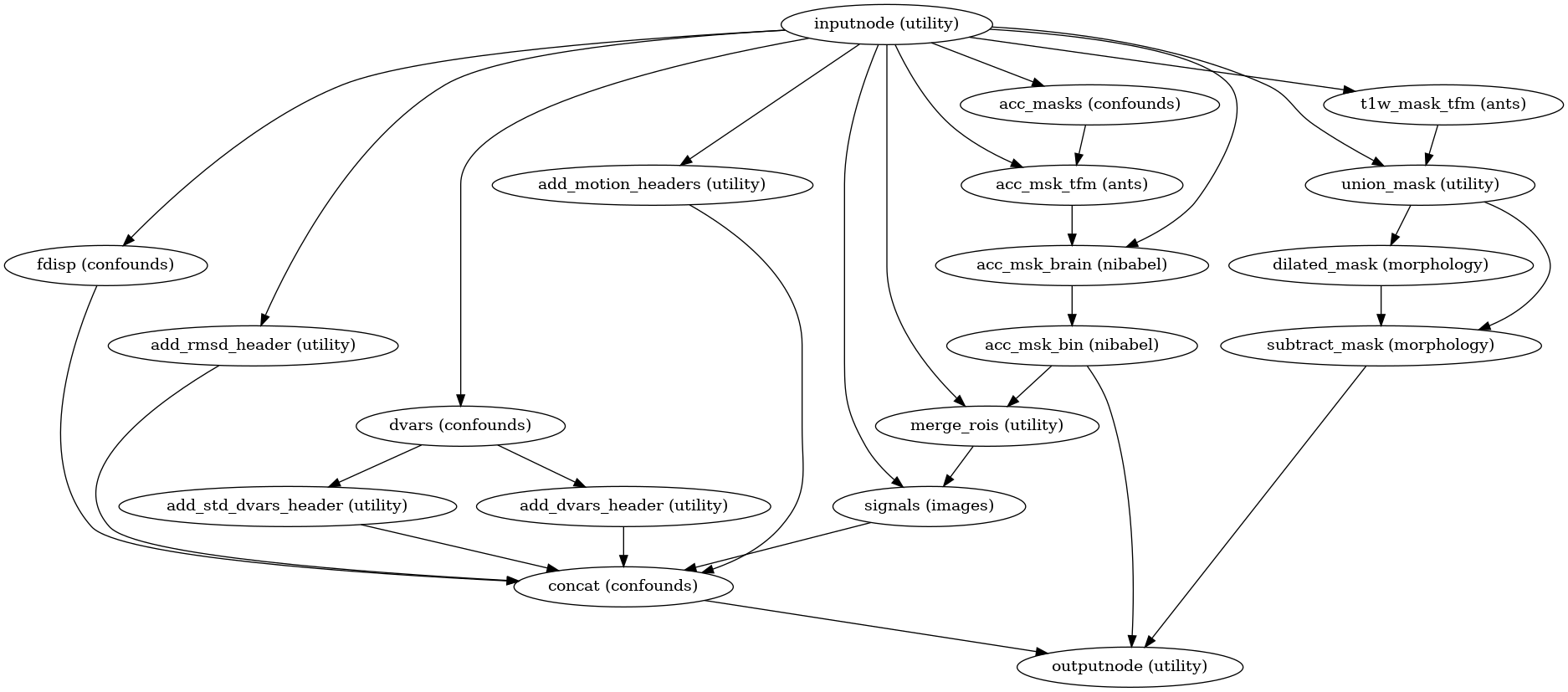
(Source code, png, svg, pdf)
Calculated confounds include framewise displacement, 6 motion parameters, and DVARS.
Susceptibility Distortion Correction (SDC)
One of the major problems that affects ASL data is the spatial distortion caused by the inhomogeneity of the field inside the scanner. Please refer to the SDCFlows documentation for details on the available workflows.
Applying susceptibility-derived distortion correction, based on fieldmap estimation
See also SDCFlows’ init_unwarp_wf() and
init_coeff2epi_wf()
Preprocessed ASL in native space
A new preproc ASL series is generated from the original data in the original space. All volumes in the ASL series are resampled in their native space by concatenating the mappings found in previous correction workflows (HMC and SDC, if executed) for a one-shot interpolation process. Interpolation uses a Lanczos kernel.
The preprocessed ASL with label and control. The signal plots above the carpet plot are framewise displacement (FD) and DVARS.
CBF Computation in native space
ASL data consist of multiple pairs of labeled and control images. ASLPrep first checks for proton density-weighted volume(s) (M0 scans). In the absence of M0 images or an M0 estimate provided in the metadata, the average of control images is used as the reference image.
After preprocessing, the pairs of labeled and control images are subtracted:
Single-Delay ASL
The CBF computation of single-delay (post labeling delay) ASL data is done using a one-compartment model [3].
(Pseudo-)Continuous ASL
For (P)CASL ([pseudo-]continuous ASL), CBF is calculated using a general kinetic model [6].
\(\tau\), \(\lambda\), \(\alpha\), and \(w\) are labeling duration, brain-blood partition coefficient, labeling efficiency, and post-labeling delay (PLD), respectively.
In the absence of any of these parameters, standard values are used based on the scan type and scanning parameters.
The element which differentiates single-delay PCASL’s CBF calculation from the PASL equivalents is \(T1_{blood} \cdot (1 - e^{\frac{ - \tau }{ T1_{blood} } })\).
Pulsed ASL
Currently, ASLPrep does not support PASL data without a bolus cut-off technique applied.
QUIPSS Modification
For pulsed ASL (PASL) data with the QUIPSS bolus cut-off technique, the formula from Wong et al.[7] is used.
where \(\Delta{TI}\) is the post-labeling delay (PLD) minus the bolus cutoff delay time.
Given that \(TI\) is equivalent to \(w\) in BIDS datasets
(i.e., as the PostLabelingDelay field),
the formula for QUIPSS is the same as PCASL,
except \(\Delta{TI}\) replaces
\(T1_{blood} \cdot (1 - e^{\frac{ - \tau }{ T1_{blood} } })\).
QUIPSS II Modification
For PASL data with the QUIPSS II bolus cut-off technique, the formula from Alsop et al.[3] is used.
where \(TI_{1}\) is the bolus cutoff delay time.
Note that the formula for QUIPSS II is the same as the one for QUIPSS, except \(TI_{1}\) replaces \(\Delta{TI}\).
Q2TIPS Modification
For PASL data with the Q2TIPS bolus cut-off technique, the formula from the commercial Q2TIPS CBF calculation is used, as described in Noguchi et al.[8].
where \(TI_{1}\) is the first bolus cutoff delay time and \(TI_{2}\) is the last bolus cutoff delay time.
Note that the formula for Q2TIPS is the same as QUIPSS II, except \(TI_{2}\) replaces \(TI\) in the numerator.
Multi-Delay ASL
In multi-delay ASL, control-label pairs are acquired for multiple post-labeling delay values. This type of acquisition requires more complicated models, but it also results in more accurate CBF estimates. Also, multi-delay ASL allows for the estimation of arterial transit time (ATT), arterial bolus arrival time (aBAT), and arterial blood volume (aBV).
For multi-delay ASL, ASLPrep uses a two-compartment general kinetic model recommended by Woods et al.[9]. The two-compartment model predicts the \(\Delta{M}\) signal as a sum of two components: a tissue compartment and an intravascular compartment. The tissue compartment reflects labeled blood water that has reached the capillary bed in the tissue at a given post-labeling delay. However, some labeled blood water will remain in arteries at that post-labeling delay. This is the intravascular compartment, which, when unaccounted for, will result in an overestimation of CBF at voxels near the arteries.
Pseudo-Continuous ASL
The tissue compartment is modeled as:
The intravascular compartment is modeled as:
For reference, please see Equations 2 and 4 in Woods et al.[9].
Pulsed ASL
The intravascular compartment is modeled as:
For reference, please see Equations 3 and 5 in Woods et al.[9].
Additional Denoising Options
For cases where data may be especially noisy (e.g., due to motion or a low-SNR protocol), ASLPrep includes options to additionally denoise CBF estimates.
The two current options are SCORE/SCRUB and BASIL.
SCORE and SCRUB
ASLPrep includes the ability to denoise CBF with SCORE and SCRUB.
Structural Correlation based Outlier Rejection (SCORE) [10] detects and discards extreme outliers in the CBF volume(s) from the CBF time series. SCORE first discards CBF volumes whose CBF within grey matter (GM) means are 2.5 standard deviations away from the median of the CBF within GM. Next, it iteratively removes volumes that are most structurally correlated to the intermediate mean CBF map unless the variance within each tissue type starts increasing (which implies an effect of white noise removal as opposed to outlier rejection).
The mean CBF after denoising by SCORE is plotted below
Computed CBF maps denoised by SCORE
After discarding extreme outlier CBF volume(s) (if present) by SCORE, SCRUB (Structural Correlation with RobUst Bayesian) uses robust Bayesian estimation of CBF using iterative reweighted least square method [11] to denoise CBF. The SCRUB algorithm is described below:
\(CBF_{t}\), \(\mu\), \(\theta\), and \(p\) equal CBF time series (after any extreme outliers are discarded by SCORE), mean CBF, ratio of temporal variance at each voxel to overall variance of all voxels, and probability tissue maps, respectively. Other variables include \(\lambda\) and \(\rho\) that represent the weighting parameter and Tukey’s bisquare function, respectively.
An example of CBF denoised by SCRUB is shown below.
Computed CBF maps denoised by SCRUB
BASIL
ASLPrep also includes the option to compute CBF using BASIL.
Bayesian Inference for Arterial Spin Labeling (BASIL) is an FSL tool for CBF estimation.
BASIL implements a simple kinetic model as described above, but uses Bayesian inference principles [12]. BASIL is mostly suitable for multi-delay ASL data. It includes bolus arrival time estimation with spatial regularization [13] and the correction of partial volume effects [14].
A sample of BASIL CBF with spatial regularization is shown below:
Computed CBF maps by BASIL
The CBF map shown below is the result of partial volume corrected CBF computed by BASIL.
Partial volume corrected CBF maps by BASIL
Quality control measures
Quality control (QC) measures such as FD (framewise displacement), coregistration, normalization index, and quality evaluation index (QEI) are included for all CBF maps. The QEI [15] evaluates the quality of the computed CBF maps considering three factors: structural similarity, spatial variability, and percentage of voxels in GM with negative CBF.
ASL and CBF to T1w registration
init_bold_reg_wf()
ASLPrep uses the FSL BBR routine to calculate the alignment between each run’s
ASL reference image and the reconstructed subject using the
gray/white matter boundary.
Animation showing ASL to T1w registration.
FSL flirt is run with the BBR cost function, using the
fast segmentation to establish the gray/white matter boundary.
After BBR is run,
the resulting affine transform will be compared to the initial transform found by flirt.
Excessive deviation will result in rejection of the BBR refinement and acceptance
of the original affine registration.
The computed CBF is registered to T1w using the transformation from ASL-T1w
registration.
Resampling ASL and CBF runs onto standard spaces
init_bold_volumetric_resample_wf()
This sub-workflow concatenates the transforms calculated upstream
(see Head-motion estimation, Susceptibility Distortion Correction (SDC))
if fieldmaps are available, and an anatomical-to-standard transform from
Structural Preprocessing to map the ASL and CBF images to the standard spaces is given by the
--output-spaces argument (see Standard and nonstandard spaces).
It also maps the T1w-based mask to each of those standard spaces.
Transforms are concatenated and applied all at once, with one interpolation (Lanczos) step, so as little information is lost as possible.